News from the Institute

On 1 December, IBC2 group leader Christian Münch took up the Lichtenberg Endowed Professorship for Molecular Systems Medicine. His binding to Goethe University Frankfurt was made possible by co-financing from four foundations: The Volkswagen Foundation is contributing a total of €2 million as part of its "Lichtenberg Endowed Professorships" program, with a further €3 million being provided by the Johanna-Quandt-Universitätsstiftung, the Alfons und Gertrud Kassel-Stiftung and the Dr. Rolf M. Schwiete Stiftung.
... (read more)
On November 27th, EMTHERA scientists enjoyed a wonderful day with Pascale Cossart from Institut Pasteur, Paris & EMBL, Heidelberg with numerous scientific discussions between our guest and our PhD candidates, postdocs and PIs. In addition, our community had the chance to listen to her lecture on "Raising a bacterium to the rank of a model system: the Listeria paradigm". The day culminated in a ceremonial event, the Dies academicus, where Pascale Cossart was awarded the honorary doctorate from the Faculty of Medicine of Goethe University Frankfurt. The day was rounded off with a dinner among colleagues in a relaxed atmosphere and a lively exchange on current research projects. We congratulate Pascale Cossart on her honor and thank her for her visit.
... (read more)
As announced today by the German Research Foundation (DFG), the Collaborative Research Centre (CRC) on selective autophagy has successfully made it into the 3rd funding period and will be funded for another four years. The CRC 1177 was established in 2016 under the lead of IBC2 Director Ivan Đikić. It is the first consortium in Germany systematically addressing several challenging questions in selective autophagy. In the previous two funding periods, research focused on mechanistic, molecular and functional aspects of selective autophagy networks and led to detailed insights into the molecular basis of autophagy, its regulation and functions of individual selective autophagy pathways. In addition, CRC 1177 became an internationally recognized technology hub for autophagy research in Germany. Together with an exceptional culture that supports independence and mentoring of young scientists, CRC 1177 attracted the most promising talents to initiate their careers in this field in Frankfurt
... (read more)
IBC2 group leader Christian Münch have been awarded with a Consolidator Grant of the European Research Council (ERC). The ERC Consolidator Grant is one of the EU's most prestigious funding programmes for individual researchers who are in an intermediate career stage after obtaining their doctorate. In addition to scientific excellence, applicants must demonstrate the groundbreaking approach of their project and its feasibility in order to receive funding.
... (read more)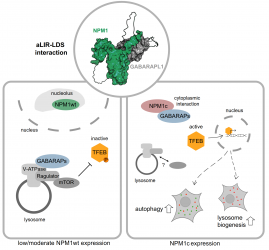
Researchers around IBC2 Vice Director Stefan Müller have uncovered a mechanism driving the accelerated growth of leukemia cells with a prevalent genetic mutation. The study focused on acute myeloid leukemia (AML), a life-threatening blood cancer prevalent in adults, with a specific mutation in the NPM1 gene. The investigation revealed that leukemia cells carrying this mutation activate specific genes vital for cellular recycling. Published in Cell Reports, the findings present promising therapeutic possibilities, offering fresh insights into combating AML.
... (read more)