News from the Institute

For the sixth year in a row, IBC2 Director Ivan Đikić is recognized for his exceptional impact by publishing multiple highly cited papers in two categories, “Biology and Biochemistry” and “Molecular Biology and Genetics”. His papers in these fields rank in the top 1% by citations based on Web of Science records from 2012 to 2022. A total of 7,125 highly cited researchers worldwide are listed this year, from which only 238 were named in two disciplines.
... (read more)
IBC2 Director Ivan Đikić is awarded an honorary doctorate from the University of Rijeka. The Senate of the University of Rijeka decided in accordance with the expert committee for the evaluation of proposals on awarding Ivan Đikić the GRADUS DOCTORIS HONORIS CAUSA for his outstanding contribution to science, scientific education and popularization of science, as well as an exceptional contribution and advices for the development of the University of Rijeka.
The honorary doctorate acceptance ceremony was held at the University of Rijeka on July 4th 2023.

This year German Association for Aging Research (DGfA) has organized the Annual meeting in Jena. By welcoming scientists from all over Germany and worldwide, the Community of Aging Researchers ensured high-quality scientific discussions, exciting talks and poster presentations. This year a major focus was on the inter-organ communication in ageing and the interplay between stress and immunity responses leading to the host metabolism reprogramming. Connecting basic science to translational ageing research and clinical trials, the conference ensured a broad overview of underlying molecular mechanisms and future therapeutic approaches in ageing-related diseases. The prize for the best oral presentation at the Annual Meeting of the German Association for Aging Research 2023 was awarded to Ines Tomaskovic for her presentation: “Gender and phenotype bias in loss-of-SPRTN-driven Progeroid Syndrome”.
... (read more)
The UCT (Universitäres Centrum für Tumorerkrankungen, University Cancer Center) science conference on 21.06.23 was organized jointly between the Philipps University of Marburg and the Goethe University of Frankfurt. It entails a diverse program in translational cancer research from basic science to clinical trials. The prize for the best oral presentation at the UCT conference was awarded to Lina Herhaus for her presentation: Tumor immune evasion through IRGQ-directed autophagy.
... (read more)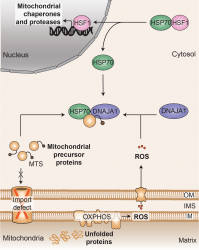
Mitochondria play a crucial role in various cellular functions, necessitating the maintenance of proper protein functionality through protein quality control pathways. When mitochondria experience protein misfolding stress, they initiate a transcriptional response to enhance the expression of mitochondrial chaperones and proteases. This process, known as the mitochondrial unfolded protein response (UPRmt), aims to restore proteostasis within the mitochondria. Although the UPRmt was discovered more than two decades ago, the exact signaling pathway by which mitochondria communicate with the nucleus to activate the UPRmt in humans has remained elusive.